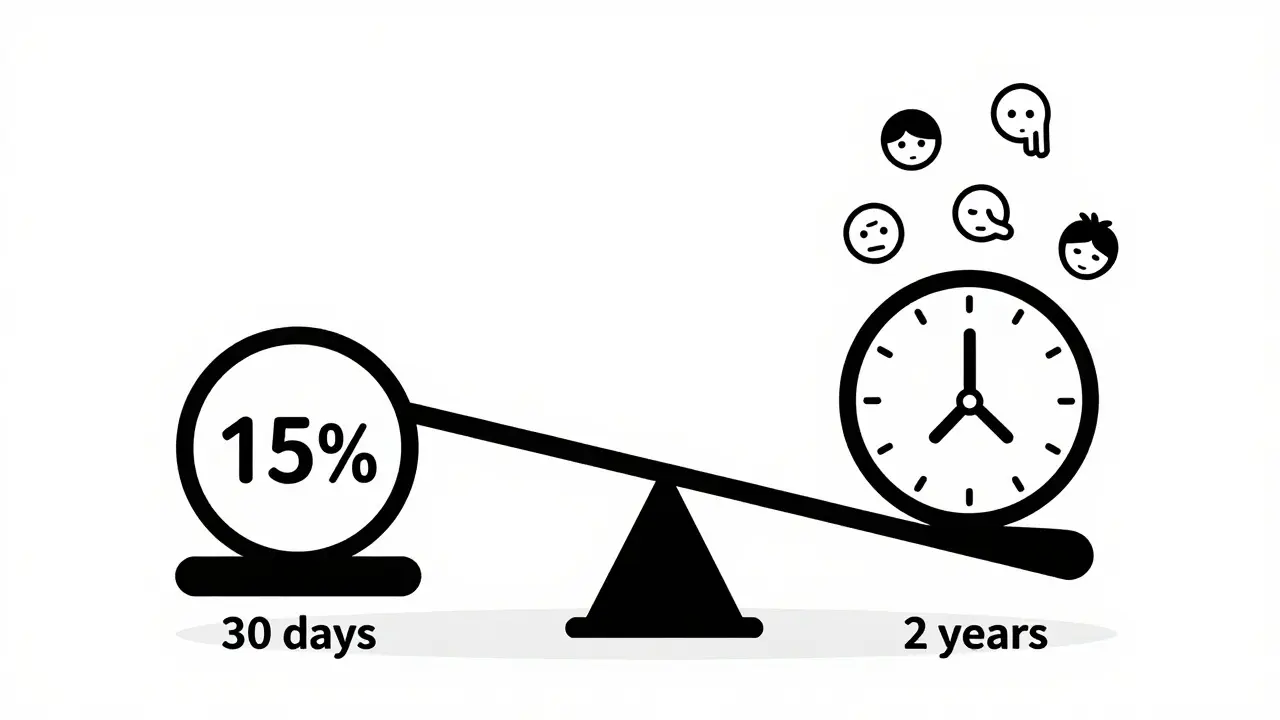
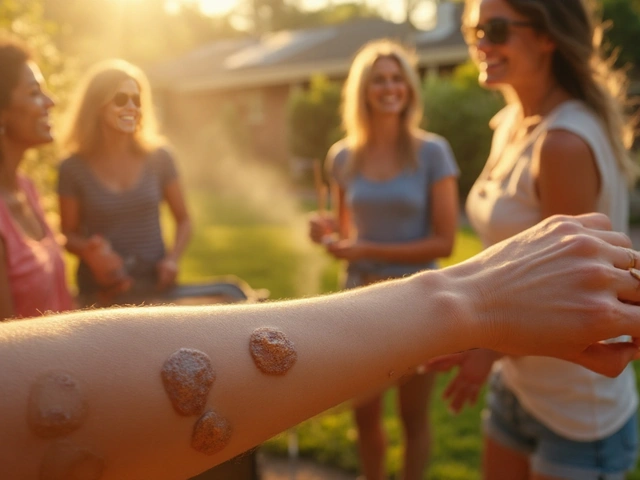Adverse event rates in clinical trials aren't just percentages-they need to account for time on treatment. Learn how the FDA now requires exposure-adjusted methods like EAIR to reveal true safety risks, and why raw percentages can mislead.
Read MoreTag: relative risk
Archive
ACA Plans and Generic Drug Coverage: How Insurance Formularies Work
Learn how ACA plans handle generic drug coverage through insurance formularies. Understand tiered pricing, cost-saving strategies, and how to maximize your benefits under the Affordable Care Act.
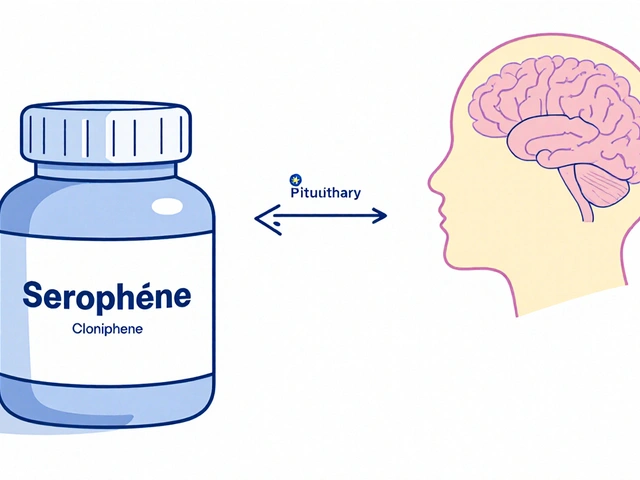
Serophene (Clomiphene) vs Alternatives: Detailed Comparison for Fertility Treatment
A thorough comparison of Serophene (Clomiphene) with letrozole, tamoxifen, gonadotropins and other fertility options, covering mechanisms, success rates, side effects, costs and practical tips.
Environmental Triggers Behind Fungal Skin Discoloration
Explore how humidity, temperature, sweat, clothing, UV light and other environmental factors fuel fungal skin discoloration and learn practical prevention tips.
Beta-Lactam Allergies: Penicillin vs Cephalosporin Reactions Explained
Most people labeled penicillin-allergic aren't truly allergic. Learn the real risks of penicillin and cephalosporin reactions, how to tell if your allergy is real, and why mislabeling costs lives and money.
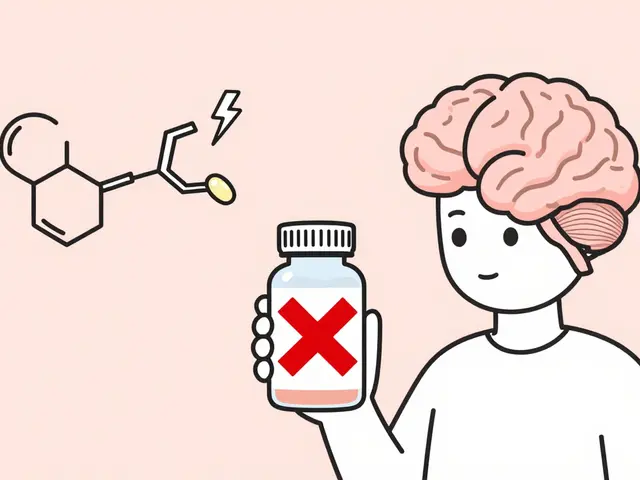
Ranitidine Explained: Uses, Risks, and Alternatives
A clear guide to ranitidine: what it does, why it was recalled, dosing basics, safety concerns, and safe alternatives for acid‑related conditions.